In 1989, the double-slit experiment revolutionized understanding of matter: The universe is not deterministic as promulgated by the worldview of Newtonian physics. Surprisingly, the meaning of quantum physics, a most arcane subject, remote from human experience, in light of these discoveries, raises the most profound question a person can ask—“Who am I?”
In January 1926, Erwin Schrödinger published in Annalen der Physik the paper “Quantisierung als Eigenwertproblem” (Quantization as an Eigenvalue Problem) that announced what is now known as the Schrödinger equation. This paper, universally acknowledged as one of the most important intellectual achievements of the twentieth century, created a revolution in physics. Quantum physics proved to be all-encompassing: For the first time, physicists understood the structure of the periodic table of chemical elements, the conduction of electricity in metals, the energy production in stars, and the first three minutes of the universe. Surprisingly, a new technology—CD players and smartphones, for instance—emerged from quantum physics, which in 1926 seemed like an esoteric branch of physics of interest to only specialists. Despite these successes, the philosophical meaning of the quantum physics revolution is still intensely debated. In January 2017, ninety-one years after the appearance of Schrödinger’s paper, Steven Weinberg, considered by many physicists as the most distinguished living member of their profession, published the essay “The Trouble with Quantum Mechanics.”[1] To understand why the meaning of quantum mechanics is still controversial, we must first see the failure of Newtonian physics to describe the atomic word.
The Abandonment of Newtonian Thinking
The success of understanding nature in terms of mechanical models led nineteenth century physicists, chemists, and biologists into the error of envisioning the future as a linear projection of the past. Many physicists thought their field was essentially complete. Addressing the British Association for the Advancement of Science, in 1900, Lord Kelvin, the greatest living English physicist at that time, predicted, “There is nothing new to be discovered in physics now. All that remains is more and more precise measurement.”[2] Albert Michelson of the famed Michelson-Morley experiment concurred: “The grand underlying principles have been firmly established . . . further truths of physics are to be looked for in the sixth place of decimals.”[3] Philipp von Jolly, an experimental physicist, advised one of his students, Max Planck, not to go into physics: “In this field, almost everything is already discovered, and all that remains is to fill a few unimportant holes.”[4]
A short time later, in 1909, in Manchester, England, Hans Geiger and Ernest Marsden under the direction of Ernest Rutherford carried out the Gold Foil Experiment to probe the structure of the atom. Rutherford expected to confirm the plum pudding model of the atom proposed by J.J. Thompson, who discovered the electron in 1897. Thompson pictured an atom where electrons (negatively-charged “plums”) move freely within a cloud of electrically positive substance (the “pudding”). Geiger and Marsden directed a beam of alpha particles, generated by the radioactive decay of radium, at a sheet of very thin gold foil. If the plum pudding model were correct, then the alpha particles would be deflected by at most a few degrees. Geiger and Marsden, however, observed that some of the alpha particles were deflected through angles much larger than 90 degrees. Rutherford was astounded: “It was quite the most incredible event that has ever happened to me in my life. It was almost as incredible as if you fired a 15-inch shell at a piece of tissue paper and it came back and hit you.”[5]
Rutherford proposed, in 1911, the planetary model of the atom, where electrons (the “planets”) orbit a positive charge concentrated in a very small volume (the “Sun”). In this model, the atom is mostly empty space. Shortly thereafter, theoretical physicists using Newtonian physics and Maxwellian electrodynamics calculated, to their dismay, that electrons continuously accelerating around the nucleus of an atom would, within a hundred-millionth of a second, lose all their energy, spiral into the nucleus, and destroy the atom. If this were true, the universe would disappear in a flash. Something was terribly wrong with the mechanical universe.
Another striking failure, labeled “the ultraviolet catastrophe,” was the prediction on Newtonian principles by Lord Rayleigh and James Jeans that a hot black body with a small hole through which electromagnetic energy could escape would produce an infinite quantity of energy—an absurd conclusion in direct contradiction to the law of the conservation of energy.
Physicist Max Planck, in 1900, after years of wrestling to understand black-body radiation, laid the foundation of quantum physics, when he proposed that electromagnetic energy could be emitted and absorbed in only discrete units, called quanta. With one simple equation, E = hf, where E is the energy element, f the frequency of the radiation, and h Planck’s action constant, he precipitated a revolution that overthrew the Newtonian understanding of matter and forged a new branch of physics—quantum mechanics.
Today, the philosophical interpretation of quantum mechanics rests on the noncontroversial, but mind-boggling, double-slit experiment.
The Double-slit Experiment
Physicist Richard Feynman taught that the double-slit experiment with single electrons is “impossible, absolutely impossible, to explain” in any way with Newtonian physics, for in it is the “heart” of quantum physics and “contains the only mystery.”[6] Like the majority of physicists in the late 1980s, Feynman believed the double-slit experiment was a gedanken (thought) experiment because the “apparatus would have to be made on an impossibly small scale to show” quantum effects.[7] The experiment, however, had already been performed in 1961 by Claus Jönsson at the University of Tubingen, and in 1974 at the University of Milan by researchers led by Pier Giorgio Merli and later in 1989 with great precision and elegance by Akira Tonomura and his colleagues at Hitachi in Japan.
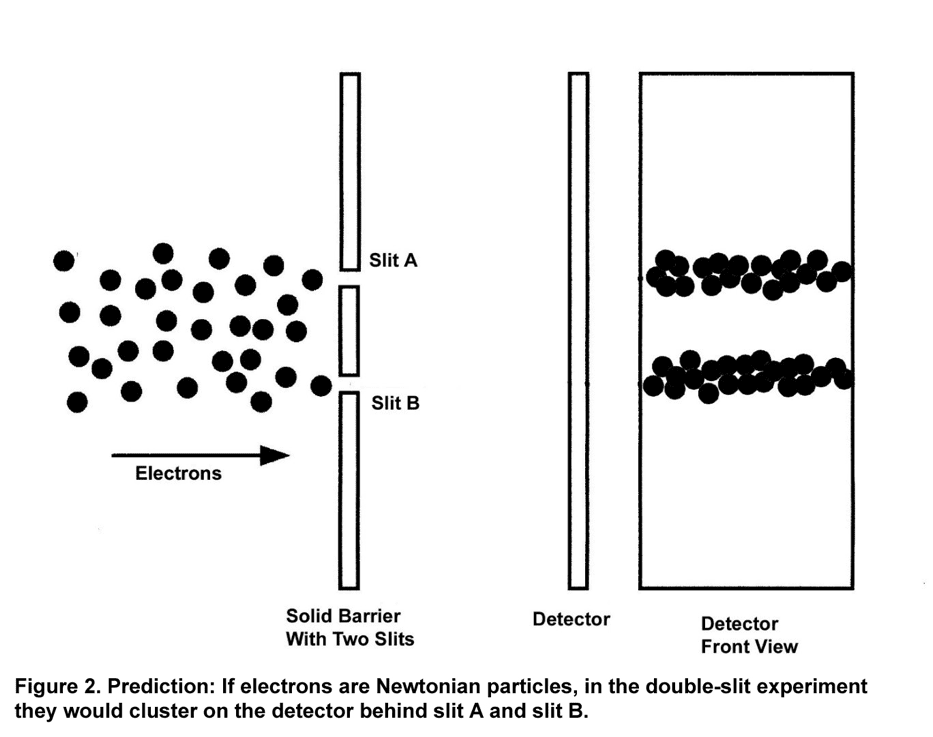
In the Japanese double-slit experiment, electrons were emitted one by one from an electron microscope.[8] They were fired in the direction of two vertical slits at the rate of 10 per second, each electron traveling with a velocity of 120,000 km/second, so that no electron could possibly interact with any other. Figure 1 is a schematic representation of the Japanese experiment; single electrons are fired one by one in the direction of two slits, labeled A and B.
If an electron were a Newtonian particle, something like a tiny baseball or a bullet, then it would necessarily follow a well-defined trajectory. Arriving at the detector, an electron would have gone through either slit A or slit B. Electrons, then, would cluster on the detector along two vertical lines behind each slit. The lines would be fuzzy, not sharp, because electrons would leave the emitter in various directions and some would ricochet off the sides of the slit openings. The distribution of electrons at the detector would be the sum of those electrons that passed through slit A and those that passed through slit B, as shown in Figure 2.
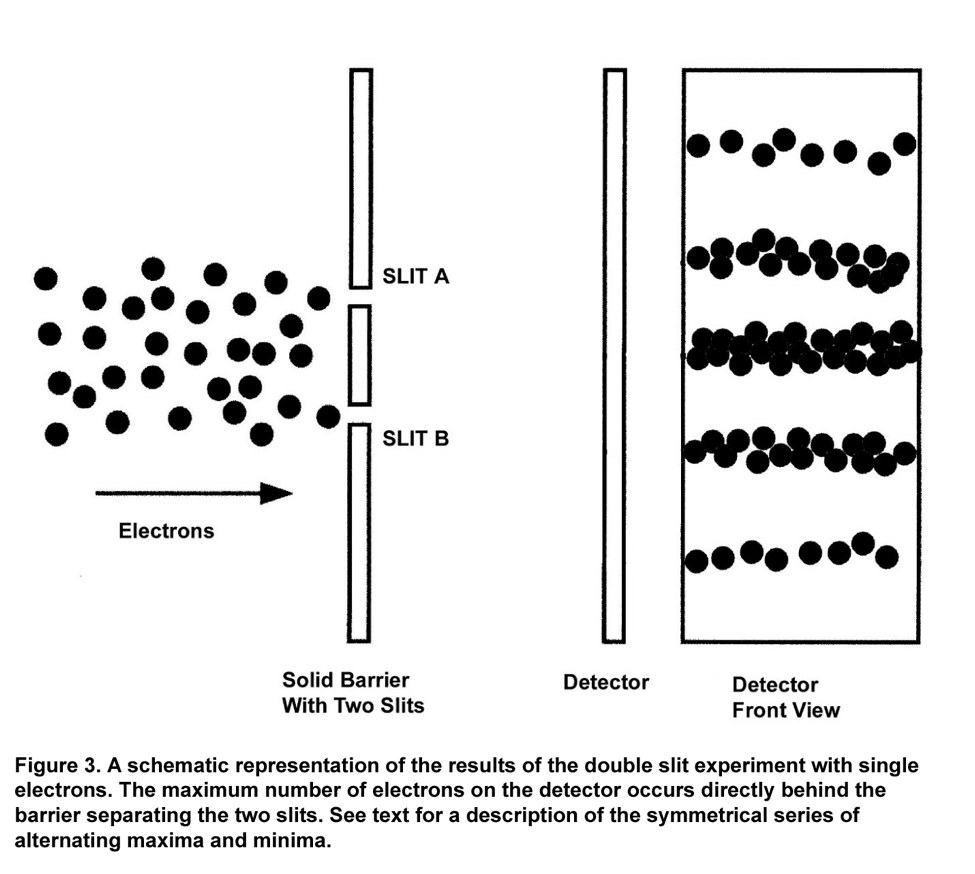
The results of the experiment were most unexpected and completely contradicted the Newtonian prediction. Figure 3 shows the number of electron hits was maximal directly behind the barrier separating the two slits! The electrons that hit the detector formed a symmetric pattern around this maximum. To the immediate sides of this maximum were two minima, places where fewer electrons struck the detector. Along the detector, a series of alternating maxima and minima occurred, with each maxima less than the preceding one.[9]
A physicist cannot say that an electron passed through either slit A or slit B, for if that were true, then the Newtonian pattern in Figure 2 would have resulted in the experiment. Unlike the bodies of Newtonian physics, where the three laws of motion keep a planet on its orbit, or direct a bullet toward its target, an electron does not follow a well-defined trajectory. In the double-slit experiment, a physicist cannot say where an electron is until it is observed at the detector.
The Japanese experimenters watched the pattern of hits on the detector slowly build up over time. At first, the electrons appeared as random hits on the detector plane, as Figure 4a shows. As electrons accumulated, a coherent pattern seemed to form. Finally, in Figure 4d, a pattern was clearly visible. Contrary to the expectation of Newtonian physics, the electrons did not cluster around two lines. Instead, one by one the electrons over time produced a pattern of alternating maxima and minima.[10]
We have reached the profound mystery of quantum physics. How is an alternating pattern of maxima and minima produced by electrons that arrive at the detector in an unpredictable manner? No one has any idea how or why this happens; physicists can just describe the results. All the fundamental equations of quantum physics—the Schrödinger and the Dirac equations among others—do not describe the behavior of individual atoms, electrons, photons, or other subatomic particles. The Schrödinger equation has nothing to say about Figure 4a and only speaks about Figure 4d. The individual electrons in the double-slit experiment arrived at the detector in a completely unpredictable manner, as shown in Figure 4a. How nature goes from Figure 4a to Figure 4d is probably a permanent mystery. According to Feynman, physicists “now believe it is impossible” to predict individual events, such as those seen in Figure 4a.[11]
We should point out a tempting error—the belief that an electron in reality must go through either slit A or slit B; the experimenter just does not know which one. But if an electron actually goes through either slit A or Slit B, then detectors at the slits can determine which slit the electron goes through and will not affect the outcome of experiment. But it can be shown that such detectors destroy the interference pattern and produce what is predicted by Newtonian physics. Thus, we must conclude that each electron in the experiment has the potential to go through either slit and this potentiality is the cause of the interference pattern. An elementary particle exists in a state of potentiality until observed. Feynman stressed that atoms, electrons, protons, indeed all elementary particles “do not behave like waves, they do not behave like particles, they do not behave like clouds, or billiard balls, or weights on springs, or like anything that you have ever seen.”[12]
In the double-slit experiment, no cause can be given why two electrons struck the detectors at different places. Individual events are unintelligible; only a large number of events, what physicists call an ensemble, exhibit mathematical regularity.[13] Stated another way, individual events are irrational, for they fall outside of reason; only a large number of events exhibit statistical regularity. The universe is not deterministic as promulgated by the worldview of Newtonian physics.
One firm conclusion of quantum physics is that all elementary particles and atoms, exist as potentialities or possibilities rather than as ordinary objects like billiard balls. Physicist Werner Heisenberg notes that atoms and elementary particles are not fully actual, but “form a world of potentialities or possibilities rather than one of things or facts.”[14] The assumption that electrons possess full existence like tiny, steel BBs contradicts the double-slit experiment. An electron, of course, is not completely indeterminate in every respect. An electron’s mass is 9.10938356×10-31 kilograms, and its charge is −1.6021766208×10-19 coulombs. These two properties are universal constants in every branch of physics and chemistry.
Atoms and elementary particles become actual only when an experimenter makes an observation. If an experimenter measures the location of an electron, he finds the electron at a particular place. If the experimenter measures the electron’s velocity, he determines its speed. But in accord with the Heisenberg Uncertainty Principle, the experimenter cannot simultaneously determine both the location and the velocity of an electron. What potentialities of the electron are actualized depends upon the observations of the physicist and thus on his or her choice of measurement strategy. Here the term “observation” always entails actualizing some aspect of the particle. Heisenberg describes how the physicist and elementary particles are related: “We can no longer talk of the behavior of the particle apart from the process of observation . . . the laws of nature which we formulate mathematically in quantum theory deal no longer with the particles themselves but with our knowledge of the elementary particles.”[15]
From the double-slit experiment, we conclude that the essential feature of quantum physics is undivided wholeness, in which the experimenter and the observing instrument is not separate from what is observed.[16] The knower and the known form an indivisible whole, as concretely seen in the Heisenberg Uncertainty Principle, to speak of the universe in the absence of any knower is absurd.
Scientific Realism
That elementary particles exist in potentiality until actualized through measurement is in direct opposition to scientific realism, the prevailing outlook of modern science from its very beginning. Galileo, in 1623, argued that tastes, odors, and colors reside only in human consciousness, and all these qualities would be wiped away and annihilated “if the living creature were removed.”[17] Reality is what is left behind when the human creature is removed. The goal of science, according to this Galilean view, is to understand nature in the absence of the scientist. The senses do not report reality; “the office of the sense shall be only to judge of the experiment, and the experiment itself shall judge of the thing.”[18] For an example of how the experiment touches nature, and the scientist touches the experiment, see Figure 5, the control room of the Tevatron.
Isaac Newton added the final two elements of scientific realism when he published, in 1687, Philosophiæ Naturalis Principia Mathematica: 1) the universe is mechanical and 2) every whole is completely understandable in terms of its smallest parts and how they interact. For the next 300 years or so, physicists, biologists, and neuroscientists attempted to prove that “the universe, including all aspects of human life, is the result of the interactions of little bits of matter.”[19]
The Revolution
In the double-slit experiment, the electrons, the measuring apparatus, and the experimenter form an undivided whole. Unlike scientific realism, in quantum mechanics the experimenter is a participant in nature as well as an observer; an understanding expressed by Bohr: “In the great drama of existence, we ourselves are both actors and spectators.”[20] Physicists have expressed this fundamental aspect of quantum mechanics in various ways. Eugene Wigner: “It was not possible to formulate the laws of quantum mechanics in a fully consistent way without reference to the consciousness.”[21] Max Born: “No description of any natural phenomenon in the atomic domain is possible without referring to the observer, not only to his velocity as in relativity, but to all his activities in performing the observation, setting up instruments, and so on.”[22] Freeman Dyson: “The laws of subatomic physics cannot even be formulated without some reference to the observer. The laws leave a place for mind in the description of every molecule.”[23]
The Rearguard
If a recent poll of 33 attendees of a conference on the foundations of quantum mechanicsis representative of all physicists, philosophers, and mathematicians, then the rearguard of the Revolution outnumbers the revolutionaries on the front line storming the citadel of scientific realism, shouting “Truth is error burnt up.”[24] Fewer than two out of five physicists hold the interpretation of quantum mechanics presented here, essentially the Copenhagen Interpretation with neo-classical holdovers, such as “wave particle duality” and “the collapse of the wave function,” stripped out. The most counterrevolutionary position adheres to the advice given to graduate physics students—“Shut up and calculate!”[25]—be mindless, do not fret over the meaning of quantum mechanics, which is philosophy, mere words.[26]
Dr. Weinberg objects to what he labels the instrumentalist interpretation of quantum mechanics (more or less the one given here) on the grounds of scientific realism. He maintains that the aim of science is “to say what is really going on out there.” By dragging humans into the fundamental laws of nature, the instrumentalist abandons the realist vision that the “world [is] governed by impersonal physical laws that control human behavior along with everything else.”[27] Yet, Dr. Weinberg does not direct us to those impersonal physical laws that control everything and that also explain quantum phenomena, for no such laws exist; the universe is not deterministic.[28]
If determinism were true, then science would be impossible. If scientists are, as their scientific outlook proclaims, nothing but a pack of neurons, and their joys, sorrows, memories, and sense of free will are in fact no more than the behavior of a vast assembly of nerve cells,then intellectual insight is an illusion.[29] If the thoughts of scientists are determined by neurophysiology, action potentials, and the endocrinology of neurotransmitters, then physics, biology, and psychology are meaningless. If every decision in science, as in ordinary life, is a “thoroughly mechanical process . . . determined by the results of prior mechanical processes,”truth is an illusion.[30]
Let me restate the logical contradiction of the deterministic outlook of science in the simplest possible terms. Suppose my interior life is determined wholly by the motions of atoms in my brain. One day, the atoms in my brain jostle around and what arises is the ancient Irish belief that leprechauns store their gold in a pot at the end of the rainbow. Such a belief clearly pertains to the atoms in my brain, not to an objective world. But this is true for all my beliefs, and hence I have no reason to suppose that any of them are true, including the one that the motions of atoms in my brain cause my interior life. From this contradiction, we can safely conclude that mind cannot be a mere byproduct of matter, and that determinism is false.[31]
The incompatibility of determinism and science has been masked over by a sleight-of-hand trick. Since Galileo, science aimed to construct a vision of the universe as if the scientist were not part of it. The physicist, the chemist, and the biologist in their imaginations watched the cosmos from the outside as if from God’s former vantage point. Determinism gives the appearance of being true through this stratagem. For example, when philosopher Bertrand Russell wrote that he looked forward to a “mathematics of human behavior as precise as the mathematics of machines,”[32] he clearly excluded himself, for he set forth his fundamental beliefs in the essay “A Free Man’s Worship.” Psychologist Joshua Greene and neurobiologist Jonathan Cohen, too, excluded themselves when they enunciated the fundamental principle of determinism: “Every [human] decision is a thoroughly mechanical process, the outcome of which is completely determined by the results of prior mechanical processes.”[33] You and I are mindless machines, but since freedom is a prerequisite for science, Drs. Greene and Cohen are not. You and I are “nothing but a pack of neurons” and our sense of free will is an illusion, but in his research program, Francis Crick, the co-discoverer of the physical structure of the DNA molecule, and his colleagues are not.[34]
The double-slit experiment and the Heisenberg Uncertainty Principle exposed the slight-of-hand trick that the scientist is not part of the universe, not included in his grand pronouncements about the cosmos. As we saw, the essential feature of quantum physics is undivided wholeness, in which the experimenter and the observing instrument is not separate from what is observed. The physicist is part of the cosmos; he cannot understand the world without including himself. Heisenberg expresses this new understanding of the scientist, “Natural science always presupposes man.”[35] Or said differently by physicist John Archibald Wheeler, “We have been promoted [by quantum physics] from observers to participators.”[36]
Currently, there are two Big Pictures of the cosmos: 1) The universe is made of parts; humans, like kangaroos and zebras, are not essential parts, only quarks and leptons are; the universe, including all aspects of human life, is the result of the interactions of little bits of matter; and 2) Things exist only in relationship and the main feature of the universe is undivided wholeness; humans, the only natural rational creatures we know of, are an essential element of the universe, and thus, the universe is composed of two constituents—mind and matter—neither reducible to the other.[37] The first Big Picture, held by most scientists and laypersons, is incompatible with quantum physics.
Surprisingly, the meaning of quantum physics, a most arcane subject, remote from human experience, raises the most profound question a person can ask—“Who am I?” Scientific realism gives one answer: “ ‘You,’ your joys and your sorrows, your memories and your ambitions, your sense of personal identity and free will, are in fact no more than the behavior of a vast assembly of nerve cells and their associated molecules.[38] You’re nothing but a pack of neurons.[39] Genes created you, body and mind; their preservation is the ultimate rationale for your existence.[40] Your desire for happiness is at loggerheads with the whole world; that you should be happy is not included in the plan of ‘Creation.’[41] You are the farcical outcome of a chain of accidents.[42] The human race is a mere chemical scum on a moderate-sized planet, orbiting around a very average star in the outer suburb of one among hundreds of billions of galaxies.[43] You are a speck of fleeting dust in an indifferent universe.”
Scientific holism inspired by quantum mechanics answers “Who am I?” in a contrary way: “You are body and mind. The causal potency of an idea, or an ideal is just as real as a molecule, a cell, or a nerve impulse.[44] Mind is the primary element of the universe. What possible sense could it make to speak of ‘the universe’ unless there was someone around to be aware of it?[45] You have the capacity to be connected to all that is.[46] Without you and other human beings, the universe would be a drama played before an empty theater and thus would be pointless.[47] Remove humankind from nature and you erase the perception of all its wonder, its beauty, and its mystery—the world becomes meaningless.”
What a surprise. The founders of the citadel of scientific realism, true revolutionaries in their day, spawned counterrevolutionaries, defenders of the modern ideology, the philosophy of materialism, which holds that every object as well as every act in the universe is matter, an aspect of matter, or produced by matter. Over ninety years have passed since the publication of the Schrödinger equation, so we can safely assume that the counterrevolutionaries are pursuing the blind hope that someday, somewhere, some brilliant physicist will understand quantum phenomena in terms of laws that control everything—a return to the deterministic outlook of Newtonian physics.
Those partisans allied to the Copenhagen Interpretation of quantum mechanics are today’s true revolutionaries, locking arms with Greek philosophers and Christian theologians, the traditional guardians of human freedom and dignity.
The Imaginative Conservative applies the principle of appreciation to the discussion of culture and politics—we approach dialogue with magnanimity rather than with mere civility. Will you help us remain a refreshing oasis in the increasingly contentious arena of modern discourse? Please consider donating now.
Endnotes:
[1] Steven Weinberg, “The Trouble with Quantum Mechanics,” The New York Review of Books (January 19, 2017).
[2] For the strange history of this quotation attributed to Lord Kelvin see “Aftermath and legacy.”
[3] Albert Michelson, quoted in “Michelson, Albert Abraham (1852-1931).”
[4] Philipp von Jolly, quoted in “Philipp von Jolly,” Wikipedia.
[5] Ernest Rutherford, quoted by David C. Cassidy, Gerald James Holton, Gerald Holton, and Floyd James Rutherford, Understanding Physics (New York: Springer, 2002), p. 632.
[6] Richard P. Feynman, The Feynman Lectures on Physics Vol. III (Reading, Mass.: Addison-Wesley, 1963), Ch. 1, p. 1. Italics in the original.
[7] Ibid., Ch. 1, p. 5.
[8] Read about the Hitachi Double-Split Experiment here.
[9] The alternating maxima and minima of the distribution of electron on the detector are called interference fringes because they resemble the decorative fringes sometimes used on clothing and rugs.
[10] See the remarkable Video Clip produced by the experimenters from their data.
[11] Ibid., Ch. 1, p. 10.
[12] Ibid., Ch. 1, p. 1.
[13] See Werner Heisenberg, Physics and Philosophy: The Revolution in Modern Science (New York: Harper & Row, 1958), p. 54.
[14] Ibid., p. 186.
[15] Werner Heisenberg, “The Representation of Nature in Contemporary Physics,” Daedalus 87 (no. 3): 99-100.
[16] Renegade physicist David Bohm, in Wholeness and the Implicate Order (London: Routledge, 1980), introduced “undivided wholeness,” which we developed in our own, specific way to indicate that the experimenter, the observing instrument, and the observed cannot be separated.
[17] Galileo, “The Assayer,” in Discoveries and Opinions of Galileo, trans. Stillman Drake (Garden City, New York: Doubleday Anchor, 1957), p. 274.
[18] Francis Bacon, The New Organon: Or the True Directions Concerning the Interpretation of Nature (Indianapolis, IN: Bobbs-Merrill, 1960 [1620]), p. 22.
[19] See H. Allen Orr, “Awaiting a New Darwin,” The New York Review of Books, 60, No. 2 (February 7, 2013).
[20] Neils Henrik Bohr, Essays 1958-1962 on Atomic Physics and Human Knowledge (New York: Wiley, 1963), p. 15.
[21] Eugene P. Wigner, Symmetries and Reflections: Scientific Essays of Eugene P. Wigner (Woodbridge, Conn.: Ox Bow Press, 1979), p. 172.
[22] Max Born, Physics in My Generation (London & New York: Pergamon, 1956), p. 48.
[23] Freeman Dyson, Disturbing the Universe (New York: Harper & Row, 1979), p. 249.
[24] Maximilian Schlosshauer, Johannes Kofler, and Anton Zeilinger, “A Snapshot of Foundational Attitudes Toward Quantum Mechanics.”
[25] To whom this phrase should be attributed to, see N. David Mermin, “Could Feynman Have Said This?”
[26] See Weinberg, “The Trouble with Quantum Mechanics.”
[27] Ibid.
[28] See George Stanciu, “Determinism: Science Commits Suicide.”
[29] Francis Crick, The Astonishing Hypothesis, (New York: Scribner’s, 1994), p. 3.
[30] Joshua Greene and Jonathan Cohen, “For the law, neuroscience changes nothing and everything,” Philosophical Transactions of the Royal Society London B (2004) 359: 1781. Available here.
[31] A similar argument was given by J.B.S. Haldane, a British geneticist and a committed Marxist. See J.B.S. Haldane, Possible Worlds: And Other Essays (London: Chatto and Windus, 1927; reprint, London: Transaction Publishers, 2002), p. 209. Page reference is to the reprint edition.
[32] Bertrand Russell, Portraits from Memory (New York: Simon & Schuster, 1956), p. 15.
[33] Greene and Cohen, p. 1781.
[34] Francis Crick, The Astonishing Hypothesis, (New York: Scribner’s, 1994), p. 3.
[35] Heisenberg, “The Representation of Nature in Contemporary Physics,” p. 100.
[36] John Archibald Wheeler, “The Universe as Home for Man,” American Scientist 62 (Nov.-Dec. 1974): 689.
[37] For a discussion of why human mind is not an epiphenomenon of matter, see George Stanciu, “Materialism: The False God of Modern Science.”
[38] Francis Crick, The Astonishing Hypothesis, (New York: Scribner’s, 1994), p. 3.
[39] Ibid.
[40] See Richard Dawkins, The Selfish Gene (New York: Oxford University Press, 1976), p. 21.
[41] Sigmund Freud, Civilization and Its Discontents, trans. and ed. James Strachey (New York: Norton, 1961), pp. 24-25.
[42] Steven Weinberg, The First Three Minutes (New York: Basic Books, 1977), p. 154.
[43] Stephen Hawking, quoted by David Deutsch, The Fabric of Reality (New York: Viking, 1997), pp. 177-178.
[44] Roger Sperry, “Mind, Brain and Humanist Values,” in New Views of the Nature of Man, ed. John R. Platt (Chicago: University of Chicago Press, 1965), pp. 78, 82.
[45] John A. Wheeler, “Genesis and Observership,” in Foundational Problems in the Special Sciences, ed. Robert E. Butts and Jaakko Hintikka (Dordrecht, Holland: Reidel, 1977), p. 18.
[46] George Stanciu, “Wonder and Love: How Scientists Neglect God and Man.”
[47] Erwin Schrödinger, Mind and Matter (Cambridge: Cambridge University Press, 1959), p. 2.
Editor’s Note: The featured image is an illustration from La Vraye Histoire du Bon Roy Alixandre (The Romance of Alexander in Old French prose) by Pseudo-Callisthenes, courtesy of Wikimedia Commons.










The author explains a great leap for science without melodramatic theology. I’m greatly impressed with the argument without claiming to be wholly a partisan of holism. To some degree we, or at least I, may have to let the mystery be.
Quantum holism became written into mechanics with the socalled energy-time uncertainty relation, which does not contradict Newton’s laws, but does rewrite Newton’s third law in terms of the quantum of action, and therefore also rewrites all three laws in terms of matrix mechanics, so that motion itself is indeterminate given that no system in motion exists only for itself.
I just want to point out that in the ancient Indian text the “Bhagavadgita” (“Song of the Lord”) there is a stanza that can be translated loosely thus: Yes, you have every right and ability to do whatever you like [say, perform an experiment] but you can never ever control the outcome”.
Secondly, there is an ancient Indian practice of propitiating the Goddess (you may have heard of the “Durga Pooja” festival widely celebrated in Calcutta) by reciting a list of her thousand names. Interspersed among names originating in mythology, magical practices, esoteric religious references, etc. are some that have profound meaning. Two examples (from the Sanskrit original) are: “Kshetra kshetrjna Paulini” – She who preserves and protects the ‘field’ and the one who knows the’field’; “Dhyaana Dhyaatru Dheya Roopaa” – She who takes the form of meditation, the meditator and that which is meditated upon.
But then does quantum mechanics suggest that the universe is entirely chaotic, that there is no real room for causality at all? Though this preserves the mystery of the universe, it eliminates the ordered beauty, and thus seems to me scarcely better than the denial of free will. Am I seeing this wrong?
Chaos theory, relativity theory, and quantum theory do not contradict Newton’s three laws of motion, but do revise his postulates on absolute time and space, hence rewrite the three laws of motion, and, mainly, relativity theory rewrites the first, chaos theory the second, and quantum theory the third law. In my humble opinion, this puts a necessary emphasis on the conscious subject: his reference frame of clocks and rods (theory of relativity), his computer (chaos theory), and his measurement apparatus (quantum theory), all of whom depend on time’s irreversibility, and therefore on Dr. Brann’s question elsewhere: What then, is time? If his study is mainly Platonic, Dr. Stanciu’s study is mainly Aristotelic. Thus, time needs not be mathematical, rather its study is that of the soul. If God is the immovable mover, Newton’s third law with quantum energy-time uncertainty allows for his grace, but this is also true for every living, animated, or rational soul, in an Aristotelian picture, with an hierarchy of consciousness. Angels mediate grace. Grace itself is probably the Holy Ghost who is charity. One must maintain that there exists both mind and matter, and that angelic time is not mathematical time.
This is the most superior introduction to Quantum Physics I have ever encountered. Clear, methodical and concise. The possible causes of the results of the double slit experiment, of which I have been familiar is particularly praiseworthy as are the various theories of the causes presented. This work should be the introduction to—at least—a layman’s book on QP, if not a chapter in a undergrad level physics text. I had hoped that you were to mention the QP interactions observed in the particles affected simultaneously while separated long distances, Einstein’s “spooky actions at a distance”. The universal connectivity (of all matter?) mentioned I believe is the source of “psychic” connectivity between humans at a distance. I recall reading about the QP experiment in which two people met in a room, interacted, then were placed in separate rooms and their brains “communicated” by choosing the identical shapes projected before them. All matter is just tumbling particles banging into each other and “all is one” as the Buddhist explain, no?
It is interesting that Albert Einstein is not mentioned in this article. Is it because he did not understand of the principles of the quantum mechanics?